SAB-142: Targeting Type 1 Diabetes (T1D) a Disease with a High Unmet Medical Need
To address a complex autoimmune disease like T1D requires a sophisticated Multi-Pathway Approach that can be administered as a single therapeutic.
T1D requires a multi-targeted, multi-epitope treatment to address the underlying abnormal immune response driving the condition.
Current treatment approaches focus primarily on daily disease management with insulin monitoring and administration to prevent complications, but these do not change the trajectory of the disease.
There is a need for a multi-targeted, disease-modifying therapies with an acceptable safety profile that can ultimately delay the onset and progression of clinical Stage 3 T1D.
SAB-142 is not another insulin nor is it for chronic disease management, it is potentially a disease-modifying therapy for T1D, envisioned to be administered once a year.
SAB-142: The First Fully-Human IgG Antibody Treatment Aimed to Provide Disease-Modifying Effects for Patients with Stage 2 or Stage 3 Type 1 Diabetes
SAB-142 is the first-in-class, fully-human alternative to rabbit- or equine-derived anti-thymocyte globulin (ATG). Its mechanism of action (MoA) is similar to that of rabbit ATG, which has been clinically validated in multiple clinical trials for T1D, demonstrating the ability to slow down disease progression in patients with new or recent onset of Stage 3 T1D.
2 Years: Low-Dose ATG* Preserved C-Peptide in New Onset T1D
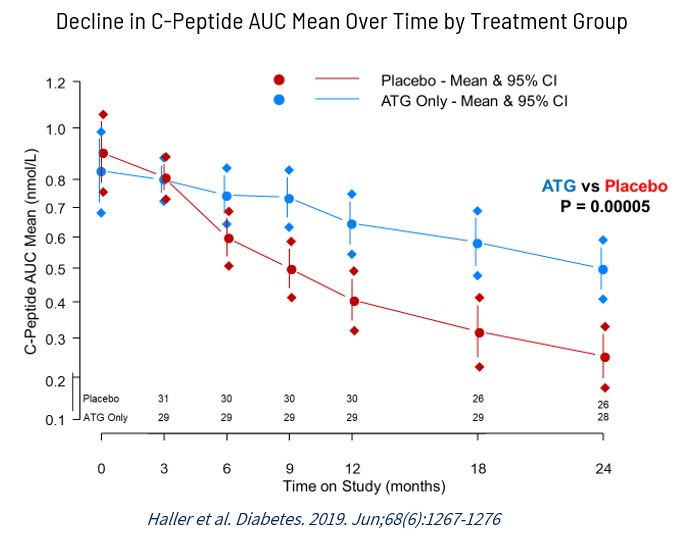
*Rabbit ATG from Sanofi – Not SAB-142 (Human TC-Bovine Derived ATG), Haller et al. Diabetes 2019. June, 68(6): 1267-1276.
Reference:
SAB Publication: “Low-Dose Anti-Thymocyte Globulin Preserves C-Peptide, Reduces HbA1c, and Increases Regulatory to Conventional T-Cell Ratios in New-Onset Type 1 Diabetes: Two Year Clinical Trial Data” Michael J. Haller, S. Alice Long, J. Lori Blanchfield, et al.
Click here to read full publication: https://diabetesjournals.org/diabetes/article/68/6/1267/39730/Low-Dose-Anti-Thymocyte-Globulin-Preserves-C
SAB-142, like rATG, directly targets multiple immune cells involved in destroying pancreatic beta cells. By stopping immune cells from attacking beta cells, this treatment preserves insulin-producing beta-cells.
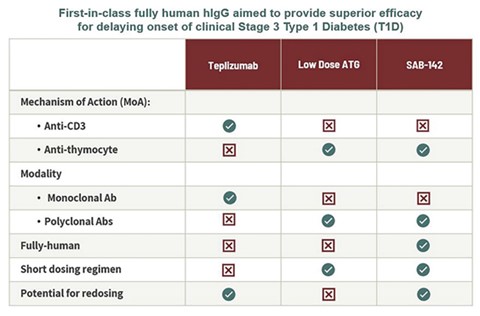
By nature of being fully-human, SAB-142 is aimed at allowing safe and consistent re-dosing for this lifelong chronic disease without the potential risk of inducing the major immune reactions that can occur with animal IgGs. Overall, SAB-142 is characterized as having a multi-target binding profile and MoA similar to FDA-approved rATG, with the potential of an improved potency and safety profile.
SAB-142: Mechanism of Action

Pathophysiology: The Biologic Basis for Disease in Adults and Children.Kathryn L. McCance, RN, PhD,Sue E. Huether, RN, PhD2014
There are multiple pathophysiology targets that need to be addressed in T1D likely required to modulate the disease. This includes modulation of “bad acting” T-lymphocytes like Cytotoxic T-cells, activated macrophages, and B-cells.

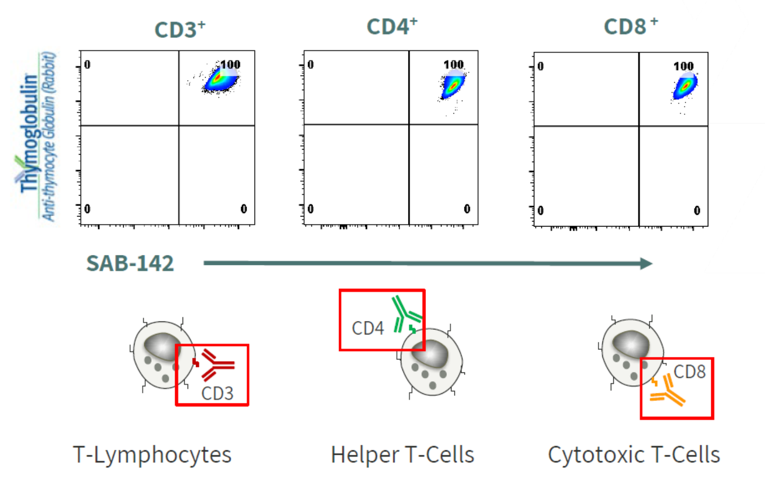
SAB-142 is demonstrated to have a similar MoA as rATG in binding to subsets of T and B lymphocytes while preserving Tregs cell populations. Similar to rATG, SAB-142 is shown to modulate multiple immune targets known to be associated with T1D.
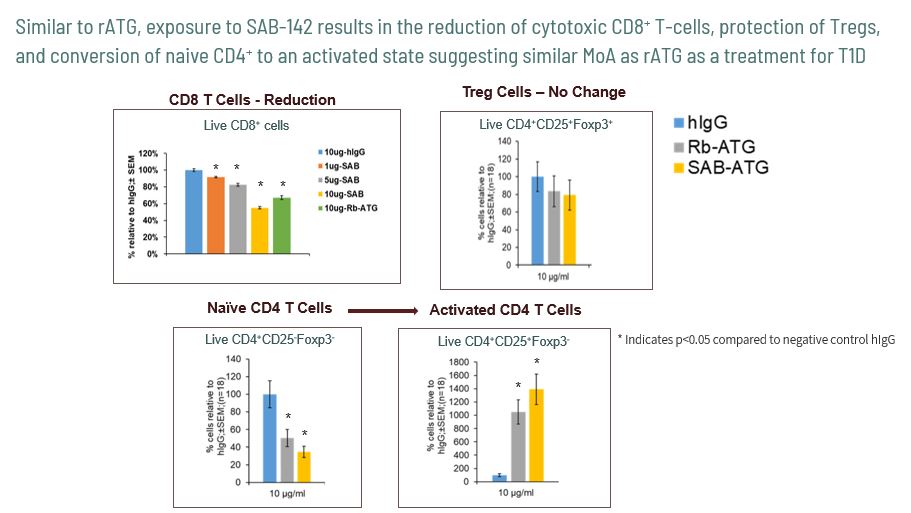
SAB-142: Anticipated Improved Safety Profile
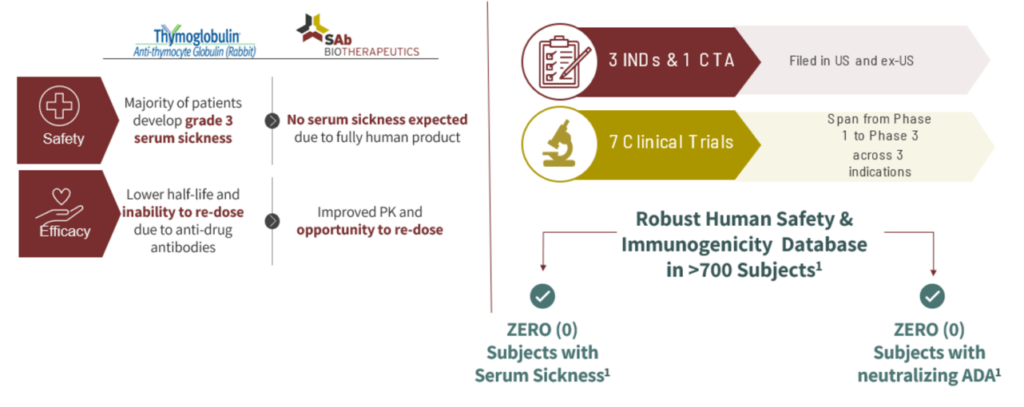
SAB-142 is anticipated to have a much-improved safety profile relative to rATG, which has significant safety concerns, as the majority of patients treated with rATG develop serum sickness from exposure to non-human, rabbit immunoglobulins.SAB’s safety and immunogenicity database in more than 700 subjects with other immunoglobulins produced by SAB’s platform show zero patients with serum sickness and zero patients with neutralizing anti-drug antibodies. This is not surprising, as SAB’s drugs are fully-human antibodies that are recognized by our immune system as native human immunoglobulins.
Status
SAB successfully completed the IND-enabling GLP toxicology study and is preparing CTA/CTN/IND submissions for Type 1 diabetes indication.
References/Additional Information
1https://www.cdc.gov/diabetes/basics/what-is-type-1-diabetes.html
2https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8304448/#:~:text=Type%201%20diabetes%20causes%20people,an%20autoimmune%20disease%20currently%20incurable.
3https://pubmed.ncbi.nlm.nih.gov/10025294/
4https://diabetes.org/diabetes/genetics-diabetes
5https://www.cdc.gov/diabetes/basics/quick-facts.html#:~:text=Type%202%20diabetes%20accounts%20for,become%20more%20overweight%20or%20obese.
6https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7146037/
7https://diabetes.org/about-us/statistics/about-diabetes
8https://www.thelancet.com/journals/landia/article/PIIS2213-8587(22)00218-2/fulltext#:~:text=In%202021%2C%20there%20were%20about,aged%2060%20years%20or%20older.
9https://www.ncbi.nlm.nih.gov/books/NBK279339/
10https://www.mayoclinic.org/diseases-conditions/type-1-diabetes/diagnosis-treatment/drc-20353017
11https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5321245/