Company Profile
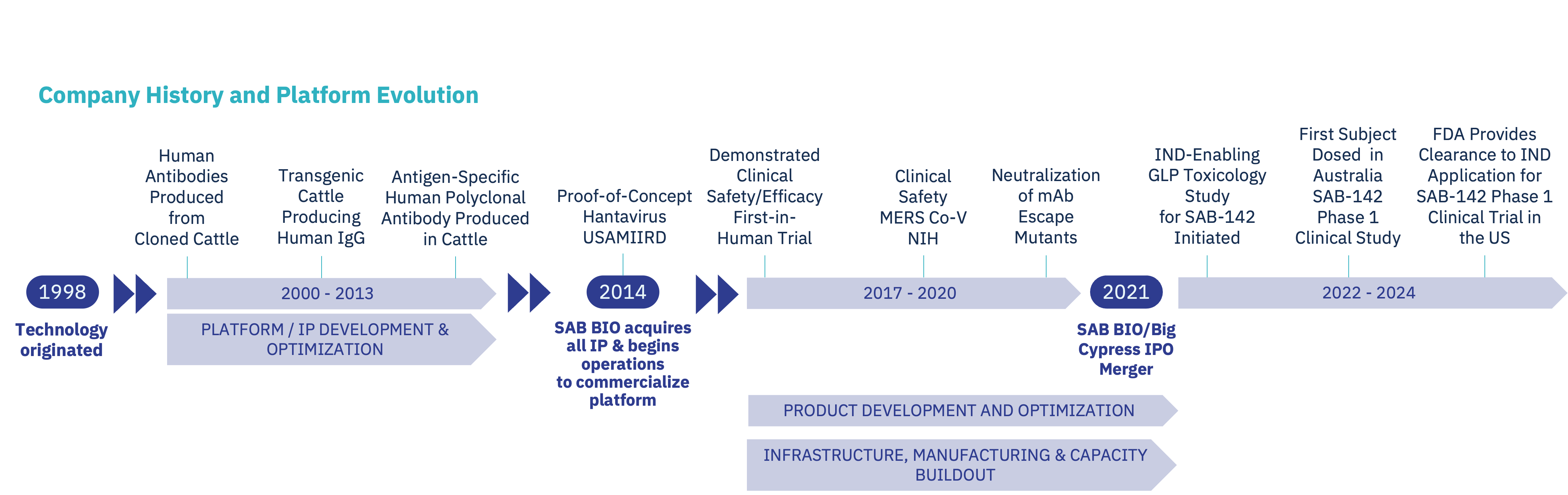
SAB BIO is a clinical-stage biopharmaceutical company working to treat and prevent immune and autoimmune disorders. Founded in 2014 and traded on NASDAQ (SABS) since 2021, SAB BIO’s innovative genetic engineering platform produces human, multi-targeted, high-potency immunoglobulins (IgGs), without the need for human donors or convalescent plasma. The science behind the platform originated in 1998, and the company began its first clinical trials in 2017.
To date, SAB BIO has conducted seven clinical trials that have dosed 700 individuals across multiple therapeutic areas. The safety and immunogenicity database for these trials shows zero patients with serum sickness or neutralizing anti-drug antibodies, which suggests an improved safety profile over other therapies.
The company’s lead investigational candidate, SAB-142, is a novel, potentially disease-modifying therapy for type 1 diabetes (T1D). As a potentially redosable human antibody, rather than an insulin or chronic therapy, SAB-142 is being investigated for its ability to delay clinical onset and progression of T1D.